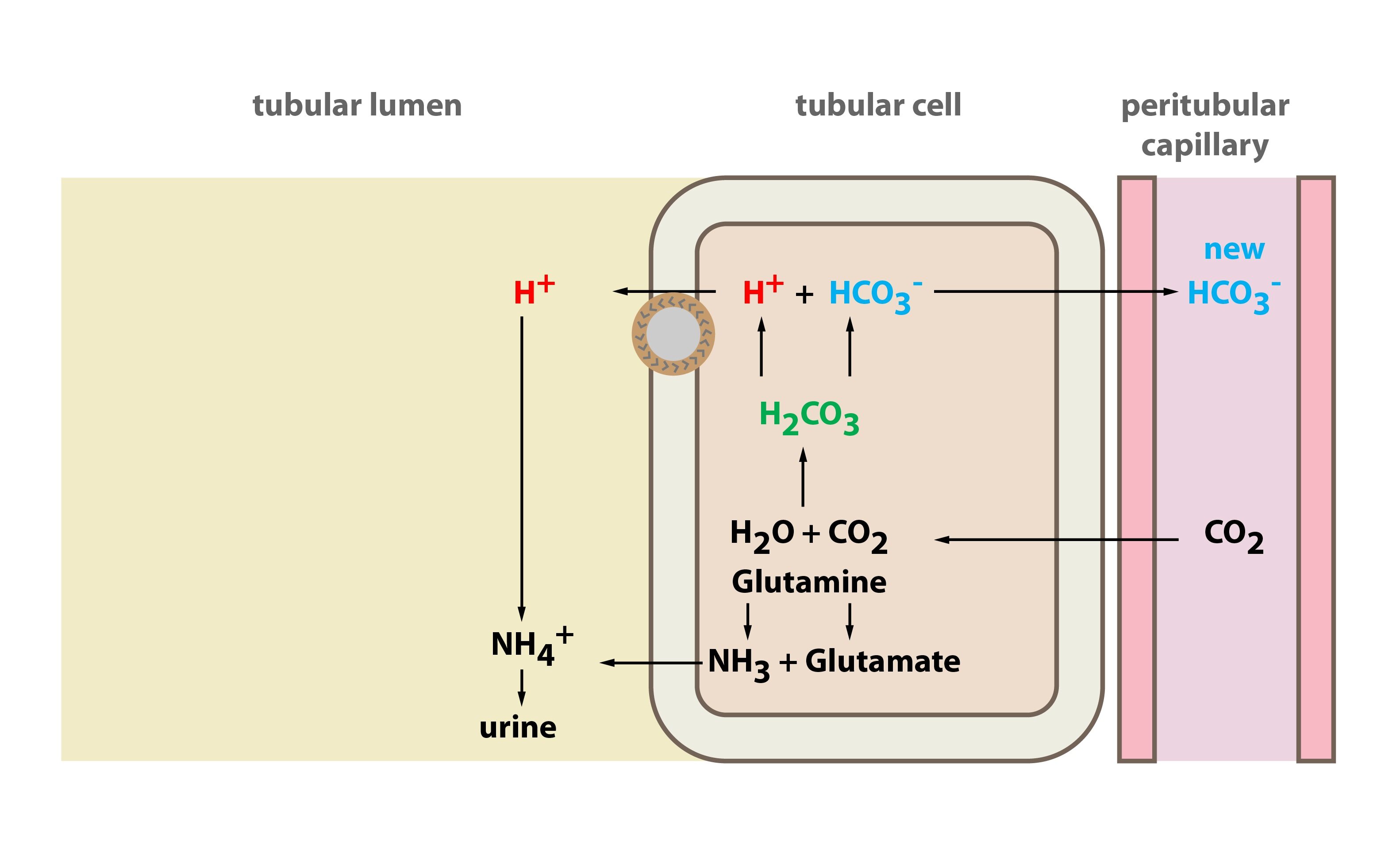
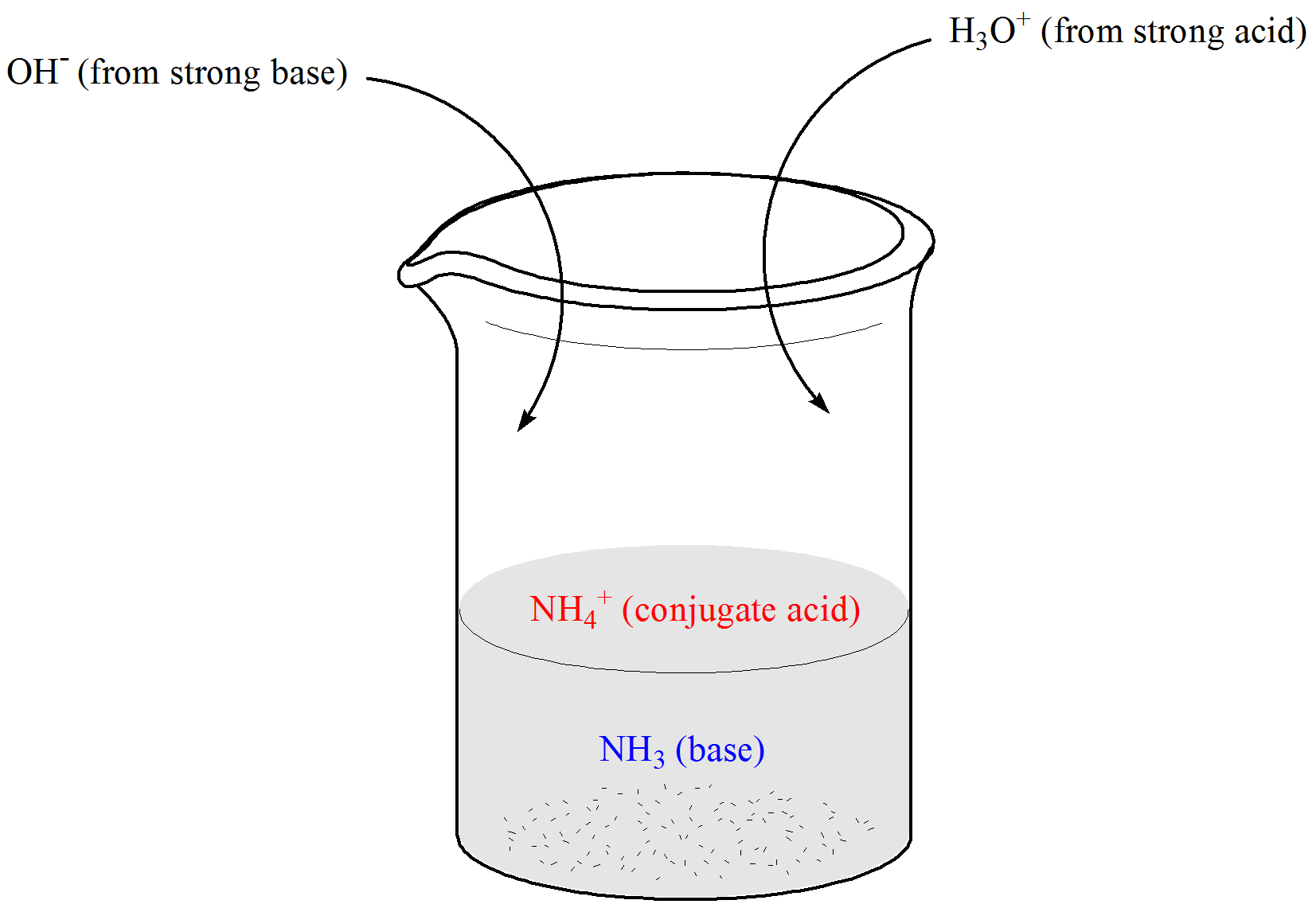
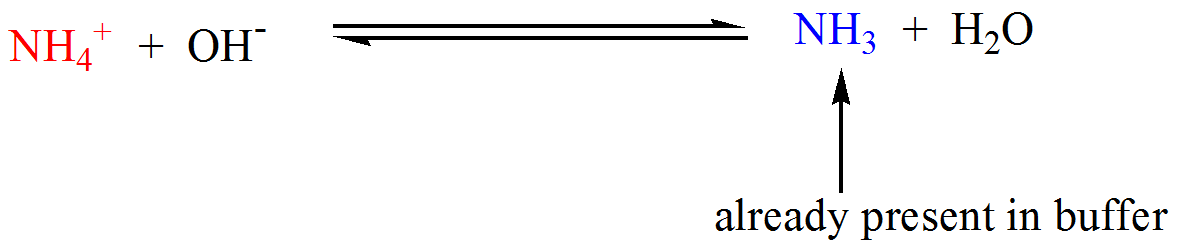
Rhesus Glycoprotein P2 (Rhp2) Is a Novel Member of the Rh Family of Ammonia Transporters Highly Expressed in Shark Kidney* - Journal of Biological Chemistry
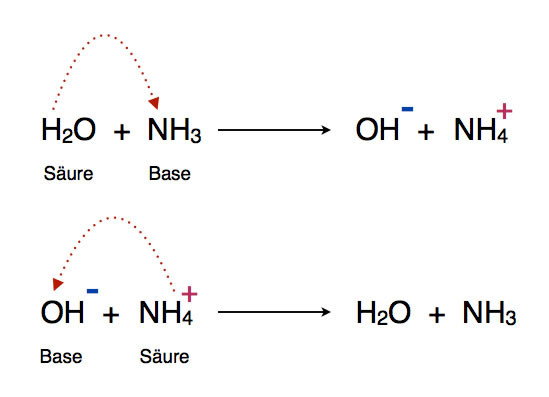
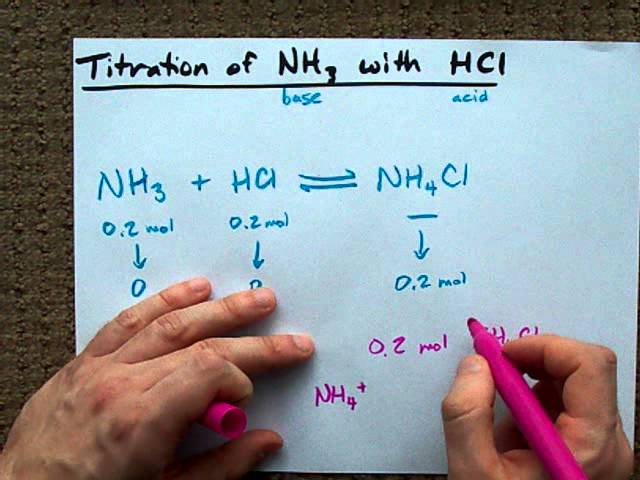
Welche Bedeutung hat die chemische Formel,,H2O+NH3- - - > OH- +NH4“ für die Säure-Base-Eigenschaften von Ammoniak? (Chemie, Säure-Base-Reaktion)

Biomedizinische Anwendungen von Calixarenen: Stand der Wissenschaft und Perspektiven - Pan - 2021 - Angewandte Chemie - Wiley Online Library
What is a “safe” level of ammonia for quarantine of fishes? | REEF2REEF Saltwater and Reef Aquarium Forum